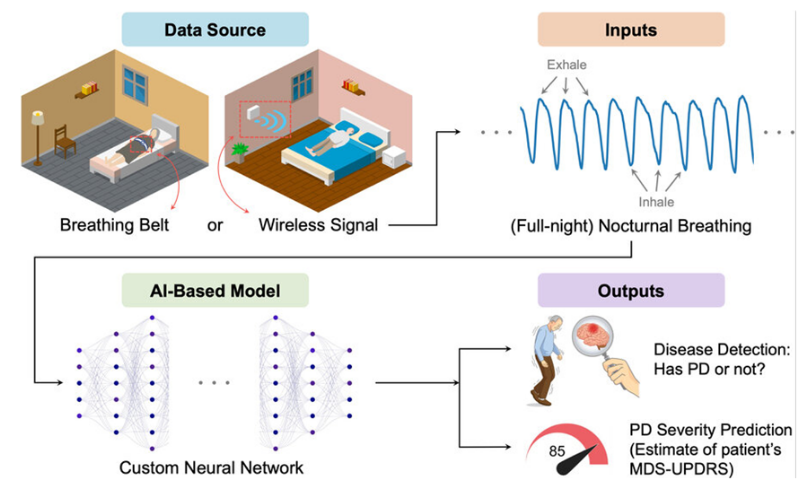
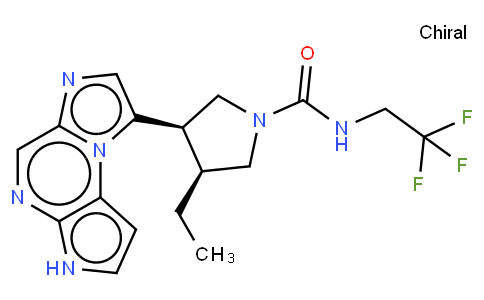
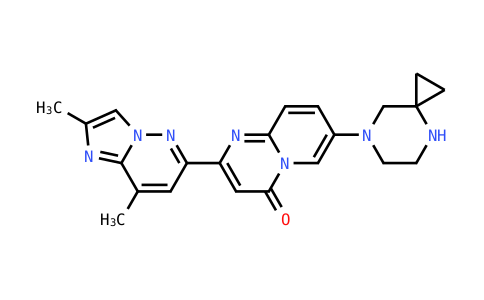
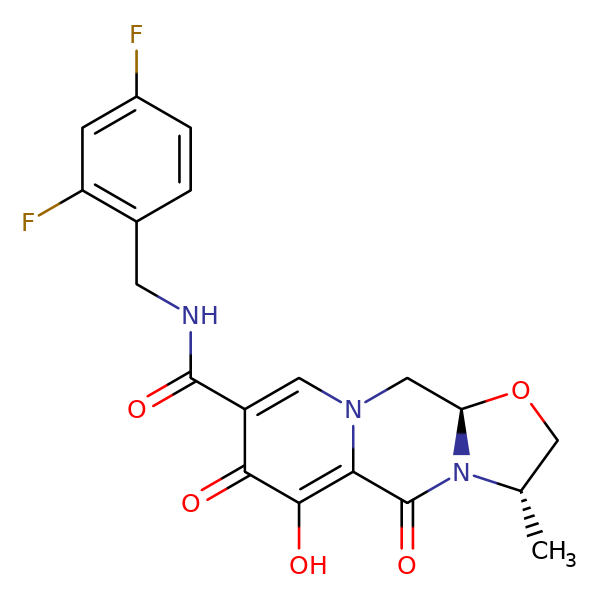
Parkinson's disease is notoriously difficult to diagnose because it relies primarily on the onse
READ MORE 2022-08-24News List
Targeting a parasite's DNA could be a more effective way to combat malaria, new research from th
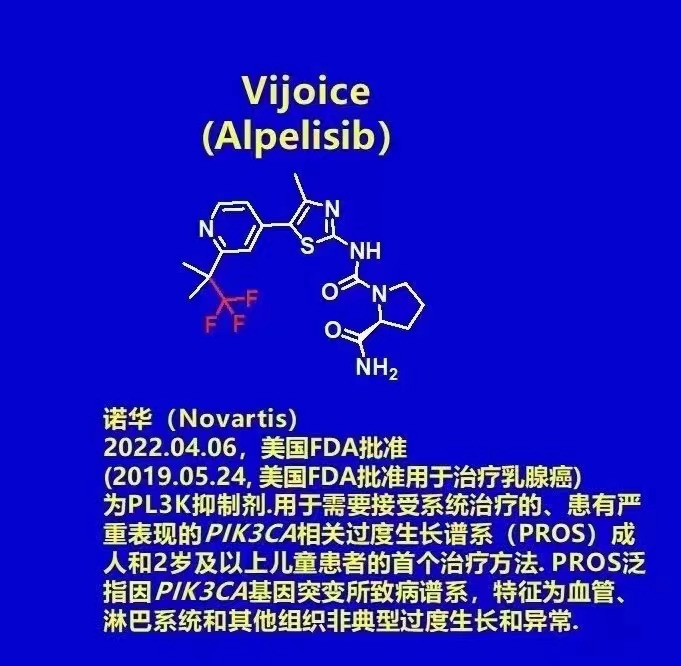
READ MORE 2022-07-282022.04.06, Novartis announced that the U.S. Food and Drug Administration (FDA) has granted accelera
READ MORE 2022-07-06Bristol-Myers Squibb (BMS) recently announced positive results from the Phase 2 PAISLEY study (NCT03
READ MORE 2022-06-13Takeda announced the Phase 3 SHP643 to evaluate the safety and pharmacokinetics of Takhzyro (lanadel
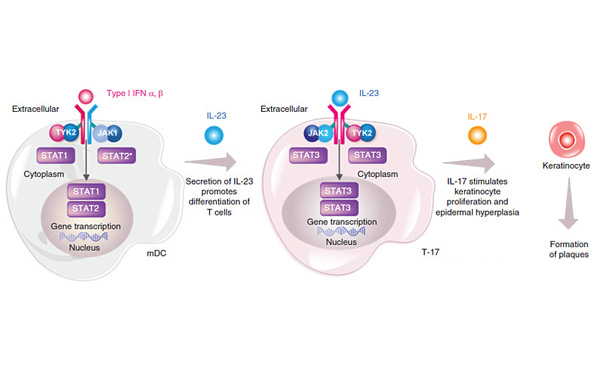
READ MORE 2022-05-13Recently, Bristol-Myers Squibb (BMS) presented 2-year data from the POETYK PSO long-term extension t
READ MORE 2022-05-12AbbVie recently announced that China’s National Medical Products Administration (NMPA) has approved
READ MORE 2022-03-11Genentech, a member of the Roche Group, today announced that the U.S. Food and Drug Administration (
READ MORE 2022-02-10ViiV Healthcare announced that the US Food and Drug Administration (FDA) approvedApretude, the first
READ MORE 2021-12-22Novartis’ asciminib hasoutperformedPfizer’s Bosulif in a phase 3 chronic myeloid leukemia (CML) stud
READ MORE 2021-11-01