Bristol-Myers Squibb (BMS) presented 2-year data from the POETYK PSO long-term extension trial of the novel oral TYK2 inhibitor deucravacitinib
2022-05-12
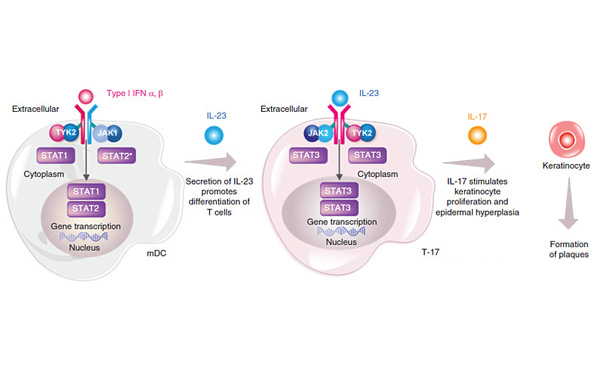
Recently, Bristol-Myers Squibb (BMS) presented 2-year data from the POETYK PSO long-term extension trial of the novel oral TYK2 inhibitor deucravacitinib in adults with moderate to severe plaque psoriasis at the Spring Symposium of the European Society of Dermatology and Venereology . The results confirmed the durable efficacy and consistent safety profile of deucravicitinib treatment.
Currently, the marketing application of deucravicitinib for the treatment of adult patients with moderate to severe plaque psoriasis is under regulatory review in multiple regions, including the United States, Europe, and Japan. If approved, deucravicitinib would be the first approved selective allosteric tyrosine kinase 2 (TYK2) inhibitor. The drug will provide patients with moderate to severe plaque psoriasis a new oral treatment option to address current care gaps.
